|
For decades, aluminum was more highly prized than gold: Napoleon III, the first president of the French Second Republic beginning in 1848, proudly served his most honored guests using aluminum plates and cutlery, because it was such a rare metal, according to The Aluminum Association. Later scientists refined the process for wresting aluminum for alum, but were unable to bring the price down to practical levels. It isn't found in pure form in nature, however in the Earth's crust, aluminum occurs most frequently as a compound called alum (potassium aluminum sulfate).ĭanish chemist Hans Christian Oersted first managed to extract aluminum from alum in 1825, according to the Thomas Jefferson National Accelerator Facility. Hsu of the Massachusetts Institute of Technology. Most common isotopes: Al-27 (stable) and Al-26 (radioactive half-life 730,000 years)Īluminum forms in stars in a fusion reaction in which magnesium picks up an extra proton, according to Chemicool, a chemistry website created by David D.Number of isotopes (atoms of the same element with a different number of neutrons): 22, one stable.Boiling Point: 4,566 degrees F (2,519 degrees C).Melting Point: 1,220.58 degrees Fahrenheit (660.32 degrees Celsius).Density: 2.70 grams per cubic centimeter.Atomic Weight (average mass of the atom): 26.9815386.Atomic Symbol (on the Periodic Table of Elements): Al.Atomic Number (number of protons in the nucleus): 13.Carbon gains oxygen to form carbon dioxide - so carbon is oxidised. In this reaction, iron(III) oxide loses oxygen to form iron - so iron(III) oxide is reduced. 
Iron(III) oxide + aluminium → iron + aluminium oxideįe 2 O 3 (s) + 2Al(s) → 2Fe(l) + Al 2 O 3 (s) The heat produced in the reaction melts the iron produced. When ignited, the mixture reacts vigorously because of the large difference in reactivity between aluminium and iron. The thermite reaction uses aluminium powder and iron(III) oxide. Sometimes, it is necessary to produce a small amount of iron more quickly, for example if railway workers need to produce molten metal to fix a broken rail. The blast furnace is used to produce iron on a large scale. In this reaction, iron oxide loses oxygen to form iron - so iron(III) oxide is reduced. Iron(III) oxide + carbon → iron + carbon dioxideĢFe 2 O 3 (s) + 3C(s) → 4Fe(l) + 3CO 2 (g) 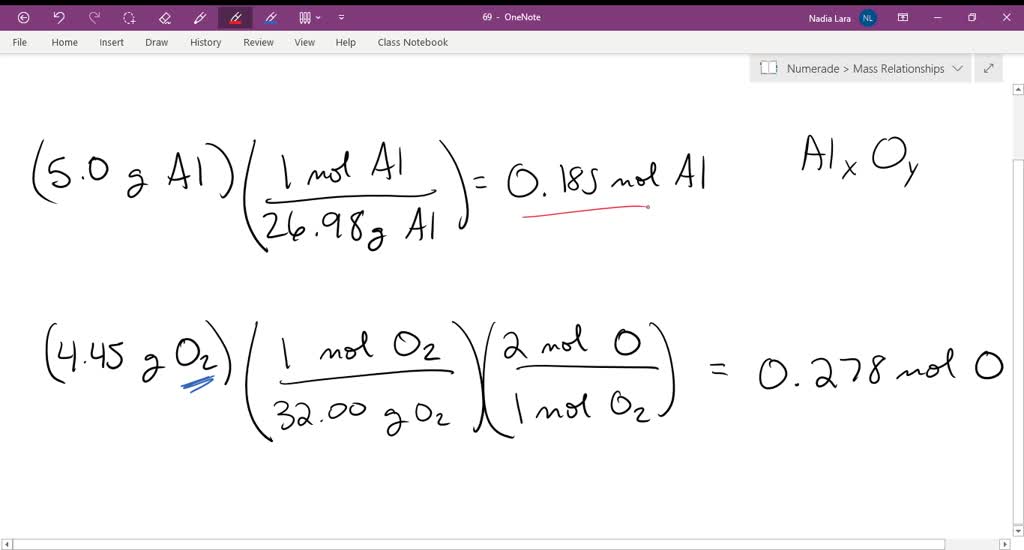
One reaction that takes place is the reaction between iron(III) oxide and carbon: Iron is extracted by heating iron ore with carbon in blast furnace. The cheapest and most efficient way of doing this is to use carbon to react with the metal oxide. Metals can be extracted from their oxides by competition reactions. Most metals are found in the form of compounds called ores. Very few metals exist uncombined in the Earth's crust. Mg(s) + CuO(s) → MgO(s) + Cu(s) Extracting metals Magnesium + copper(II) oxide → magnesium oxide + copper It can remove oxygen from copper(II) oxide: For example, magnesium is more reactive than copper. The mixture needs to be heated together in order to react. 
Metals and competition reactions Competition reactionsĪ more reactive metal can remove oxygen from the oxide of a less reactive metal.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
